Explore, Enhance, Engage
There is no one-size-fits-all approach to third party risk management. Ethixbase360 offers configurable options proportionate to risk exposure, no matter where you are in your business journey.
Why Ethixbase360?
A third-party risk management platform and methodology configured to your business needs
Ethixbase360 supports your business’ third-party anti-bribery & corruption, modern slavery, and ESG risk management with scalable platform & modular point solutions. Our Explore, Enhance and Engage methodology enables companies to manage their value chain based on risk exposure from low-touch automated screening across the entire network to enhanced due diligence and hands-on engagement with high-risk suppliers and intermediaries.
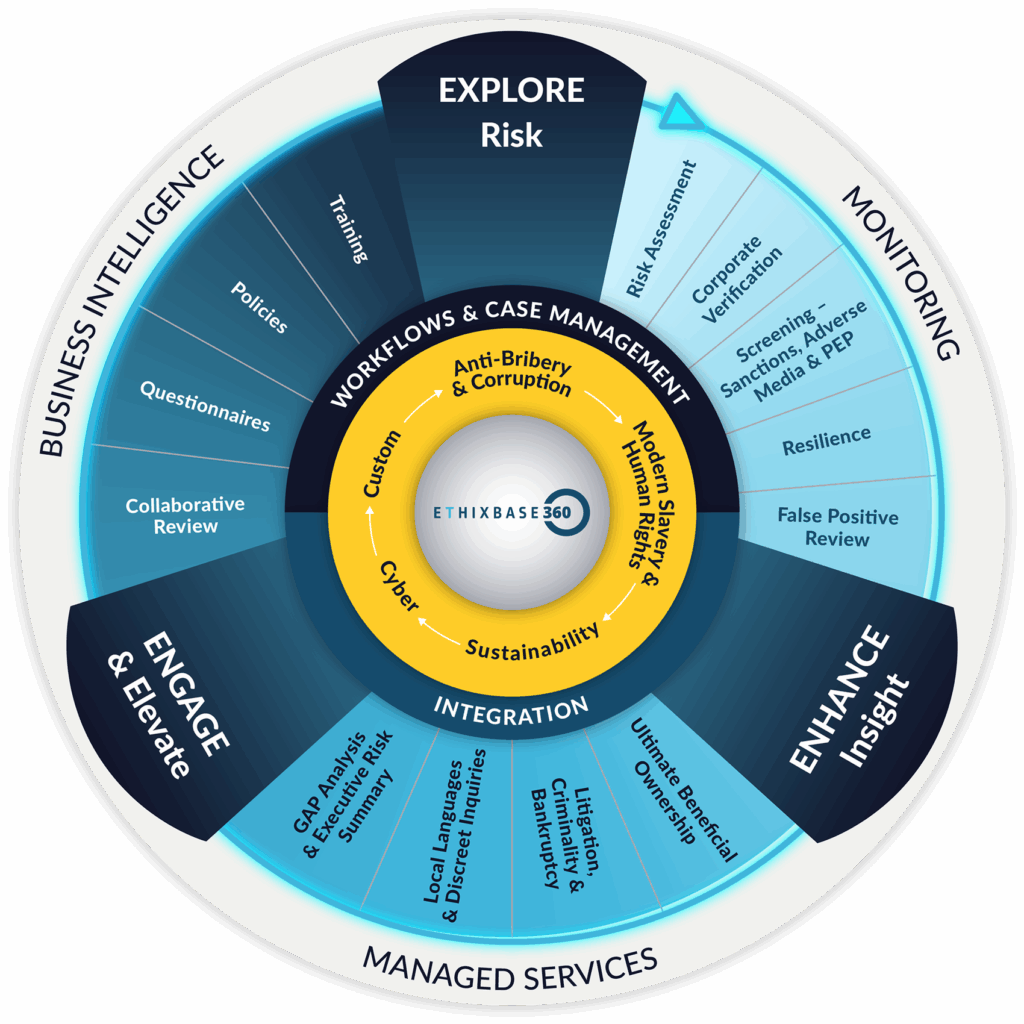
EXPLORE Risk & Resilience
Ethixbase360’s unique Explore, Enhance, Engage methodology was designed to help you take a complete approach to third party risk management across your value chain. Our EXPLORE solutions allow you to take rapid, cost-effective and configured approach to assessing third parties – identifying where resources are best allocated.
Check out our EXPLORE Platform Guide to learn more.
ENHANCE Insight with Enhanced Due Diligence
When you need to know more, Ethixbase360’s Enhanced Due Diligence reports offer a risk-based approach to deep-dive intelligence. Third parties are reviewed by our analysts located in 7 strategically located research operation centres with 40 languages in-house. Research is drawn from publicly available and ethically sourced materials with varying scopes of research available dependant on your business needs.
Check out our ENHANCE Platform Guide to learn more.
ENGAGE Directly to Elevate Standards
Elevate standards, facilitate transparency, and promote principled performance among third parties by engaging directly on key areas of concern and opportunities for improvement. The Ethixbase360 suite of Engagement solutions provide risk specific assessments to support compliance with specific regulation in addition to opportunities to collaborate with third parties to increase transparency and evidence compliance.
Check out our ENGAGE Platform Guide to learn more.
MAXIMIZE TPRM PROGRAM INVESTMENT
Ethixbase360’s configurable technology allows you to automate your third-party risk management workflows based on existing processes, with the ability to scale, refine and evolve as your business grows in size and complexity.
Leverage Ethixbase360’s team of experienced analysts to get the most out of your TPRM program. With in-house dedicated managed service teams for questionnaire follow up, screening remediation and classification available.
Integrate Ethixbase360 into existing workflows and internal systems via our RESTful API, to streamline third party onboarding, ongoing management and integrate actionable intelligence.
How it Works, by Risk Area
A third party risk management platform configured to your unique ABAC program. With an end-to-end set of ABAC platform capabilities tailored to your business needs.
Assess risk and demonstrate
compliance to stakeholders and
regulators with robust year on year reporting while combatting modern slavery risks in your
supply chain.
Explore ESG risk and resilience across your complete value chain to uncover third party vulnerabilities and engage directly to elevate standards.
Ready to get started?
Let’s talk. One of our representatives can help you shape your project.
Resources
Our Latest News, Case Studies, Webinars & More

Compliance Implications of Anthropic’s Dispute with the Pentagon
In late February 2026, the US Department of Defense (DoD) designated the artificial intelligence (AI) company Anthropic a supply chain risk, a first for a US company. [...]
Human Rights Due Diligence and Modern Slavery: Global Expectations for Compliance Programs
Human rights and modern slavery risks are receiving heightened regulatory and enforcement attention across global supply chains.[...]
Key Takeaways from our Webinar: Managing Human Rights and Corruption Risks Across Life Sciences Third Parties
Key Takeaways from our Webinar: Managing Human Rights and Corruption Risks Across Life Sciences Third Parties In Partnership with Ethics and Compliance Switzerland. [...]
Anti-Corruption, Fraud & Global Compliance For Life Sciences
We’re proud to sponsor 21st Annual Anti-Corruption, Fraud & Global Compliance for Life Sciences conference on May 5–6, 2026! They’ll be sharing how Ethixbase360 supports life sciences companies[...]
Ownership Transparency and Sanctions Risk: Building a Defensible UBO Program
Ultimate beneficial ownership (UBO) has moved from a background AML requirement to a central control for sanctions compliance, export controls, and third-party risk management. [...]
Human Rights Due Diligence and Modern Slavery: APAC and Global Compliance Expectations
Human rights and modern slavery risks are receiving increasing regulatory and enforcement attention across global supply chains. Governments are moving beyond transparency-led approaches toward clearer expectations that companies.[...]Build Trust
Ethixbase360 is committed to keeping your data safe and that journey starts with our commitment to privacy and compliance.



